|
“Valence electrons are electrons in the outer shells that are not filled.” Valence electrons are involved in the majority of chemical reactions because they have more energy than electrons in inner orbits. The number of electrons that an atom must lose or gain in order to achieve the nearest noble gas or inert gas electronic configuration is referred to as valence. These electrons are known as valence electrons. Only the electrons in the outermost shell are capable of participating in the formation of a chemical bond or molecule. Not all electrons, however, are associated with the atom. Furthermore, these electrons are in charge of atom interaction and the formation of chemical bonds. Valence electrons are all negatively charged particles that are arranged in various orbitals or shells. The tracking of valence electrons and the prediction of bond types are both aided by Lewis structures. When we study and observe an element’s atom, we come across tiny subatomic particles known as valence electrons. 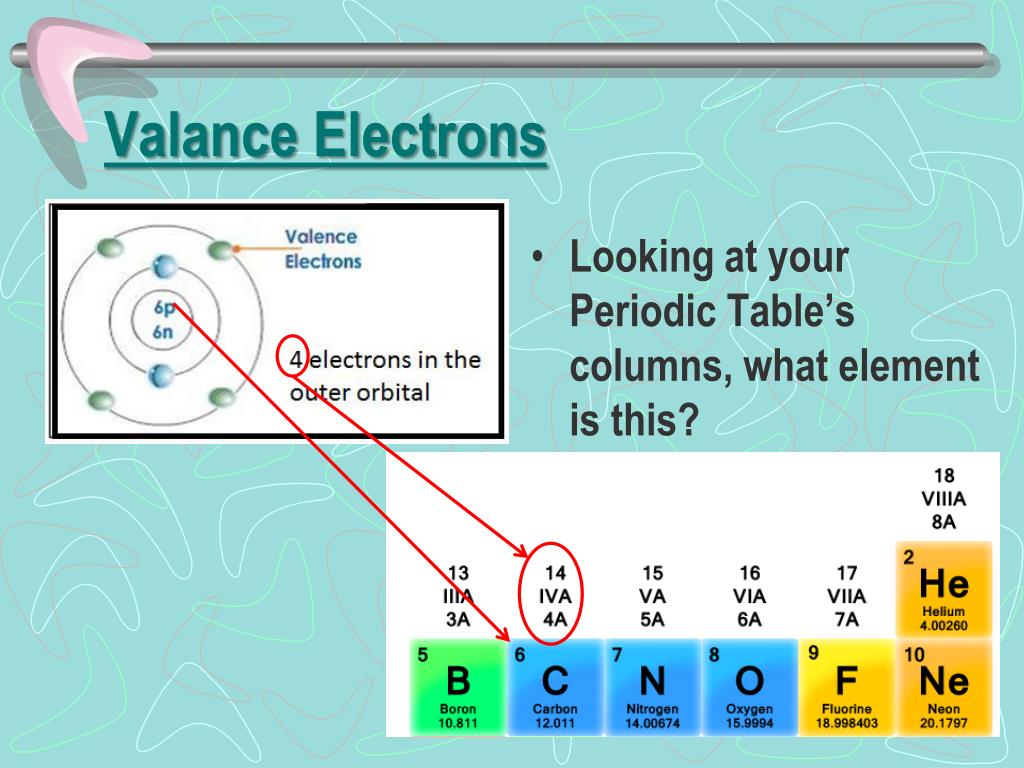
The electrons in the inner shell are known as core electrons. The s and p electrons in the outermost shell are valence electrons. Valence electrons are electrons found in an atom’s outermost shell because the electrons in the outermost shells of two atoms are the first to come into contact with each other and are the ones that determine how an atom reacts in a chemical reaction when two atoms interact. When an electron loses energy (and thus emits a photon), it can move to an inner shell that is not completely occupied. Alternatively, the electron can break free from the shell of its associated atom, resulting in the formation of a positive ion. An increase in energy can cause an electron to move (jump) to an outer shell, a process known as atomic excitation. A valence electron, like a core electron, can absorb or release energy in the form of a photon. An atom with one or two electrons fewer than a closed shell is reactive because of its proclivity to either gain the missing valence electrons and form a negative ion or to build a covalent connection by sharing valence electrons. 
Atoms with one or two extra valence electrons than a closed shell are highly reactive because the energy required to remove the extra valence electrons to form a positive ion is relatively low. A valence electron can only exist in the outermost electron shell of a main-group element a valence electron can also exist in an inner shell of a transition metal.Ī chemically inert atom has a closed shell of valence electrons (corresponding to a noble gas configuration). In this sense, an element’s reactivity is significantly influenced by its electrical arrangement. The presence of valence electrons can influence an element’s chemical properties, such as its valence-whether it can bond with other elements and, if so, how easily and how many times. A valence electron is an electron linked with an atom in the outer shell that can participate in the formation of a chemical bond if the outer shell is not closed. Click on 'Element Atomic Number', 'Element Symbol', 'Element Name' and 'Element Valence Electrons' headers to sort.In a single covalent bond, both atoms contribute one valence electron to form a shared pair. This Valence Electrons table gives the Valence Electrons of all the elements of periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
